The World Health Organization (WHO) recommends that children in Sub-Saharan Africa and other places with moderate to high Plasmodium falciparum malaria transmission get the RTS,S /AS01 (RTS,S) malaria vaccine. The proposal is based on the outcomes of a trial program that has touched over 800,000 children in Ghana, Kenya, and Malawi since 2019.
The mortality rate of malaria is over 500,000 people each year in Africa, with about half of them being children. The new vaccine isn’t flawless, but experts believe it will help turn the tide.
“This is a historic moment. The long-awaited malaria vaccine for children is a breakthrough for science, child health and malaria control,” said WHO Director-General Dr Tedros Adhanom Ghebreyesus. “Using this vaccine on top of existing tools to prevent malaria could save tens of thousands of young lives each year.”
Malaria kills over half a million people per year, almost entirely in Sub-Saharan Africa, including 260,000 children under the age of five. GlaxoSmithKline’s new vaccine stimulates a child’s immune system to fight Plasmodium falciparum, the deadliest of the five malaria infections and the most common in Africa.
The vaccine was approved by the World Health Organization on Wednesday, the first stage in a procedure that should lead to widespread distribution in low-income countries.
Having a safe, moderately effective, and ready-to-distribute malaria vaccine is “a historic event,” said Dr. Pedro Alonso, director of the World Health Organization’s Global Malaria Programme.
The vaccine, also known as Mosquirix, is not only a novelty against malaria, but also the first to be developed against any parasitic disease. Parasites are much more complex than viruses or bacteria, and the search for a malaria vaccine has been going on for a hundred years.
“It’s a huge jump from the science perspective to have a first-generation vaccine against a human parasite,” Dr. Alonso said.
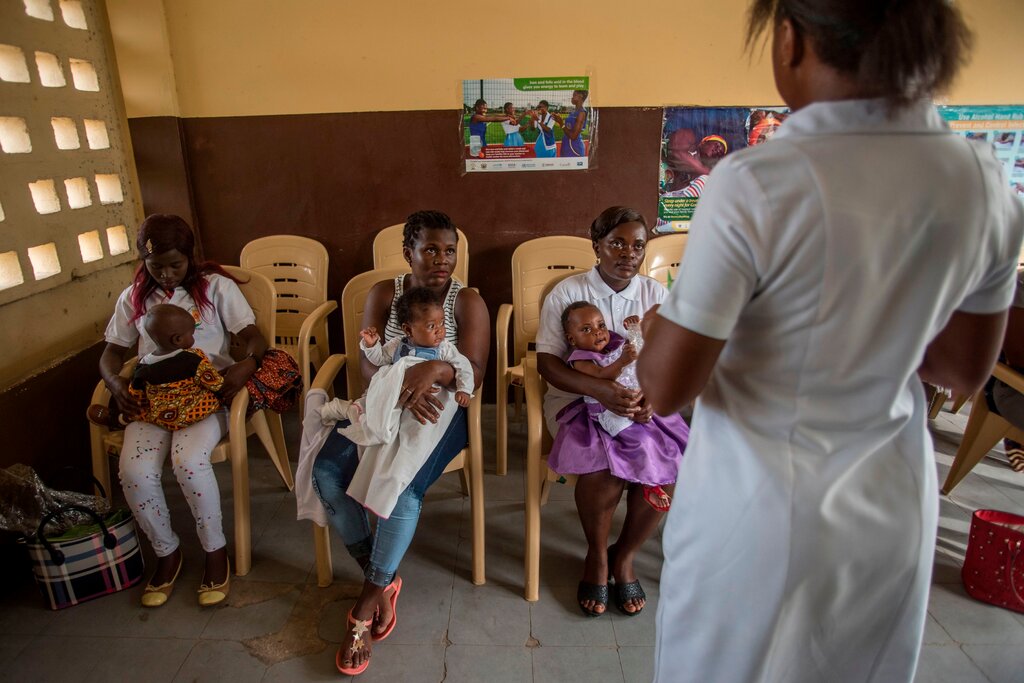
The vaccine, RTS,S, produced by UK pharmaceutical firm GSK, reduced hospitalizations for severe malaria by 30% in a pilot study in Ghana, Kenya, and Malawi. Between 2001 and 2015, the Bill and Melinda Gates Foundation provided catalytic financing for the late-stage development of RTS,S.
The RTS,S vaccine is the first and only one to demonstrate such promising outcomes, but because it must be given in three doses, one month apart, with a fourth dose a year later, the WHO needed to observe how it functioned in a “real-world” context before approving it. The pilot study will be continued in the three pilot nations in order to determine the additional value of the fourth vaccination dose and to assess the long-term impact on child mortality.
A recent study revealed that using the vaccine in conjunction with preventative medicines given to children during high-transmission seasons was far more successful than using either strategy alone in preventing severe illness, hospitalization, and death.
In the context of comprehensive malaria control, WHO advises that the RTS,S/AS01 malaria vaccine be administered to prevent P. falciparum malaria in children residing in WHO-defined moderate to high transmission areas. The RTS,S/AS01 malaria vaccine should be given to children starting at the age of 5 months on a four-dose regimen to reduce malaria illness and burden.
Sources: WHO
Sky news
The New York Times